-
Home
-
About JCTR
-
Gold Open Access
-
Issues
-
Editorial board
-
Author guidelines
-
Publication fees
-
Online first
-
Special issues
-
News
-
Publication ethics
-
Partners
-
Submit your manuscript
-
Submit your review report
-
Editorial Office
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. ISSN print: 2382-6533 ISSN online: 2424-810X
Volume 8 Issue 2
Human stem cells prevent flap necrosis in preclinical animal models: A systematic review
Francisco R. Avila, Ricardo A. Torres-Guzman, María T. Huayllani, Gunel Guliyeva, Abba C. Zubair, Alfredo Quiñones-Hinojosa, Antonio J. Forte*, Rachel Sarabia-Estrada
Avila et al. J Clin Transl Res 2022; 8(2):6
Published online: March 1, 2022
Abstract
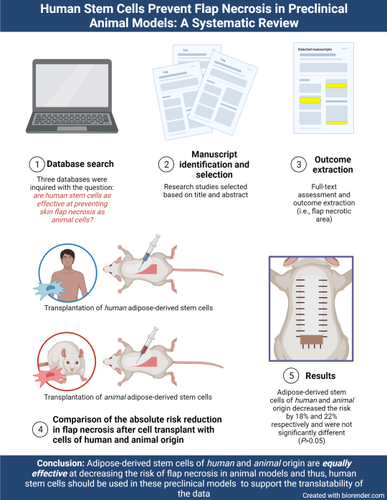
Background and aim: Adipose-derived mesenchymal stem cells (ADSC) have been proven effective to prevent distal skin flap necrosis in preclinical models. However, to appropriately translate these findings to clinical trials, the effect of ADSC of human origin (hADSC) needs to be evaluated. We hypothesize that hADSC treatment is as effective as animal ADSC treatment at preventing distal skin flap necrosis in animal flap models.
Methods: Three databases were inquired on August 17, 2020, to evaluate the necrotic flap area after using hADSCs in animal models of ischemic flaps. No publication status or dates were considered. Studies were included if they used hADSCs, measured the surviving or necrotic skin area of flaps, used animal models, and were in English. Studies were excluded if they did not use cells of human origin. The flap survival or necrotic area, perfusion, capillary density, and VEGF and HIF-1α secretion or expression were extracted.
Results: Ten studies met inclusion criteria. The mean absolute risk reduction (ARR) in necrotic skin area was 22.37% (95% confidence interval [CI] 16.98% - 27.76%, P<0.05) for flaps treated with animal ADSCs and 18.04% (95% CI 2.74% - 33.33%, P<0.05) for flaps treated with hADSCs. The difference between mean ARRs was not statistically significant (4.33%, 95% CI -34.47% - 43.13%, P>0.05).
Conclusion: Human ADSCs prevent skin flap necrosis to the same degree as animal ADSCs in rodent and rabbit flap models.
Relevance for patients: This review found that adipose-derived stem cells of human origin are equally effective at reducing the risk of surgical flap necrosis in preclinical models of small animals as autologous animal cells. The findings in this review should encourage researchers to use human adipose-derived stem cells in animal models of ischemic flaps to accelerate their translation into clinical trials and, eventually, surgical practice. The low immunogenicity of these cells should be leveraged to gain insight into the effects of the products that will be ultimately administered to patients. Furthermore, human adipose-derived stem cells’ pro-angiogenic mechanism of action sets this therapy as a promising preventive measure for flap necrosis.
DOI: http://dx.doi.org/10.18053/jctres.08.202202.006
Author affiliation
1. Division of Plastic Surgery, Mayo Clinic, Jacksonville, Florida.
2. Department of Laboratory Medicine and Pathology, Mayo Clinic, Jacksonville, Florida
3. Department of Neurosurgery, Mayo Clinic, Jacksonville, Florida.
*Corresponding author
Antonio J. Forte
Mayo Clinic Florida, 4500 San Pablo Rd, Jacksonville, FL 32224
Tel: 904-953-2073
Email: ajvforte@yahoo.com.br
Handling editor:
Michal Heger
Department of Pharmaceutics, Utrecht University, the Netherlands
Department of Pharmaceutics, Jiaxing University Medical College, Zhejiang, China

